Phytoplankton Community Project
Guest post from Nicole Schanke of the DiTullio lab at the College of Charleston
The DiTullio lab at the College of Charleston partnered with Charleston Waterkeeper during their 2021 sampling season. The goal of this collaboration was to investigate the phytoplankton community within the Charleston Harbor area and the various environmental factors (i.e. temperature, salinity, nutrient availability) that might impact phytoplankton biomass and taxonomic diversity. We have a specific interest in the cyanobacteria community, as this phytoplankton taxon can thrive in warm, brackish, coastal waters. Some cyanobacteria species have also been shown to produce cyanotoxins that can impact human health.
Our analysis of the Waterkeeper samples included chlorophyll a, pigment and nutrient concentrations, bacterial abundance, temperature and salinity. Together, these data provide insight into the abundance and composition of the phytoplankton community, the nutrients available to support primary production and the seasonal and tidal changes in temperature and salinity that the Charleston Harbor experiences. We are currently processing samples to determine phytoplankton community composition and bacterial abundance. Herein, we present data on phytoplankton biomass and phosphate concentrations. Preliminary data are presented from three weeks of sampling (July 14th, August 11th and September 22nd).
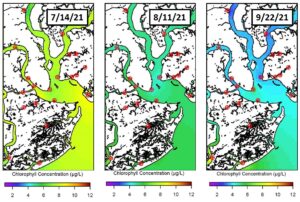
Chlorophyll a concentrations are a useful proxy for phytoplankton biomass and were plotted throughout the Charleston Harbor area for the weeks of July 14th, August 11th and September 22nd. Here, chlorophyll a concentrations were observed to be the highest on July 14th, with concentrations decreasing in August and September. Across these three weeks, the locations with high phytoplankton biomass vary. On July 14th, high biomass was found within the harbor, specifically near the outflows of Cross Creek and James Island Creek. However, on August 11th and September 22nd, the locations with the highest biomass were found near Folly Beach and the Stono River. Typically, the center of Charleston Harbor and northward to the Wando River and Filbin Creek had lower biomass.
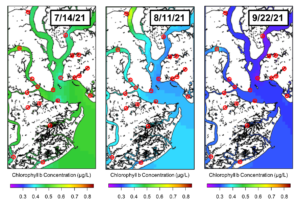
This figure displays the concentration of chlorophyll b, a diagnostic pigment of chlorophytes, a type of green algae. Again, we see chlorophyll b concentrations decrease across these three weeks, with the sites at AR3 and FC1 having higher concentrations than other locations.
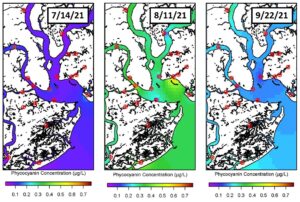
While chlorophyll a is the major photosynthetic pigment in cyanobacteria, this taxon also contains phycocyanin, a pigment‑protein complex, that serves as a proxy for cyanobacteria biomass. Phycocyanin concentrations in the Charleston Harbor were plotted from the same three weeks of sampling (July 14th, August 11th and September 22nd). Here, we observed relatively low cyanobacteria biomass on July 14th with higher concentrations on August 11th September 22nd. Of these three weeks, the highest cyanobacteria biomass was observed on August 11th, especially near Shem Creek and the mouth of the Ashley River. However, during the same week, relatively low cyanobacteria biomass was observed in the center of the harbor and in the Wando River. These variations in cyanobacteria biomass over a small spatial scale are intriguing and warrant further exploration.
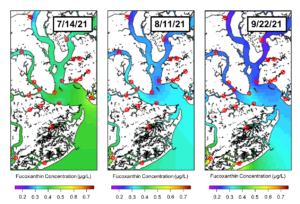
This figure displays the concentration of fucoxanthin, a diagnostic pigment of diatoms. Here, we see a spatial pattern over these three weeks, with higher fucoxanthin concentrations being observed in the Charleston Harbor and lower concentrations in the rivers.
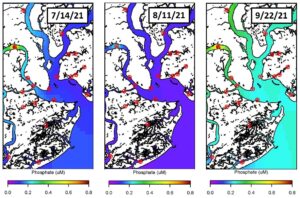
This figure displays the concentration of phosphate, a macronutrient important to phytoplankton. High nutrient concentrations, especially high ratios of phosphate to nitrate, can sometimes trigger cyanobacterial blooms relative to other phytoplankton species in coastal waters. Phosphate concentrations from the same three weeks of sampling (July 14th, August 11th and September 22nd) were also plotted throughout the Charleston Harbor area. As phosphate is an important macronutrient for phytoplankton growth (especially cyanobacteria), it is logical that areas with high phosphate concentrations would support higher phytoplankton biomass. As a general trend, the AR3 site, located up the Ashley River, had the greatest phosphate concentrations throughout the entire sampling season. However, at this location, only moderate to low chlorophyll a concentrations were measured. This suggests that phosphate may not have as strong as an influence on phytoplankton biomass as originally expected and that other environmental factors (i.e. nitrate concentrations) may more tightly control the phytoplankton community in the Charleston Harbor.
As we continue to process samples and analyze data, we will hopefully be able to better explain the Charleston Harbor system and its impact on the phytoplankton community. We will also be able to better describe the phytoplankton community composition, which, preliminarily, appears to be more diverse than expected.
We greatly appreciate the work of Charleston Waterkeeper in helping our lab explore the local waters, and we look forward to building on this dataset in the upcoming years.
